Market Trends: Pharmaceutical packaging
Pharmaceutical packaging: Getting ahead of the competition
New U.S. and European Union regulations, competition and other challenges force market players to keep moving forward.

Print & Check Flex serialization unit by Antares Vision offers flexibility, high performance.
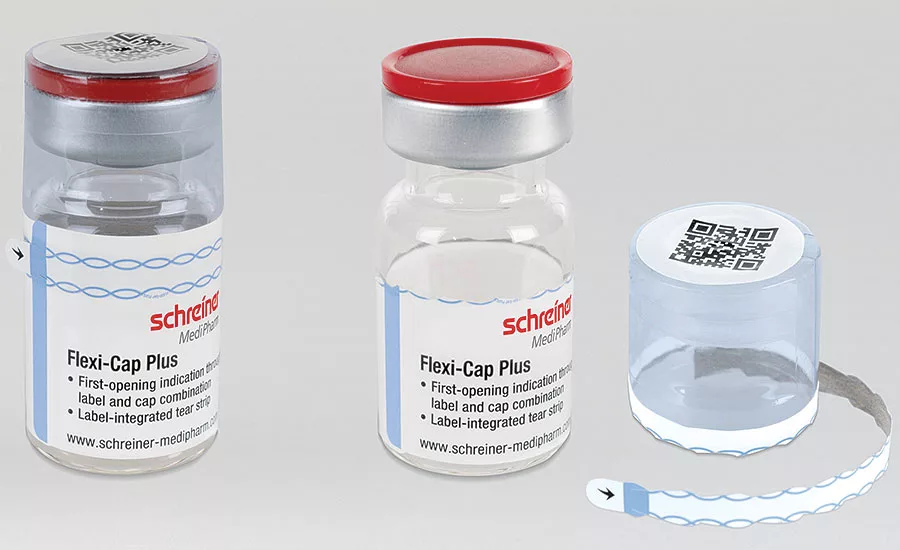
Schreiner Medipharm’s Flexi-Cap Plus uses a label-integrated tear strip for added security.
The pharmaceutical packaging market is constantly faced with new challenges and fierce competition. The stakes are high, as are the risks companies will take. With global pharmaceutical sales crossing the $1 trillion mark, according to the 2015 CMR International Pharmaceutical R&D Executive Summary, pharmaceutical packaging is more competitive than ever.
Factors contributing to market growth include demand of advanced pharmaceutical packaging solutions, such as anti-counterfeiting and serialization solutions, and both U.S. and international regulations, according to Pharmaceutical Packaging Market - Forecasts from 2016 to 2021 by Research and Markets. New products, increasing demand for blister packaging, online printing technology and investment in R&D will also drive the market.
Serialization/Track-and-Trace Solutions
Serialization is a hot topic lately. With track and trace mandates in the U.S. and European Union looming, companies need to act fast to avoid being out of compliance with new, stricter regulations. Safety and security are top of mind when it comes to incorporating a comprehensive solution.
The Print & Check Flex serialization/track-and-trace system by Antares Vision (antaresvision.com) was created for packagers and brand owners running a wide range of formats on the same lines and requiring flexibility in terms of carton dimensions, frequent artwork changes and varying code-printing specifications.
This latest addition to the series of standalone/in-line modules, prints fixed and variable data on cartons via inkjet or laser techniques and then checks the accuracy and integrity of the printed data. The Print & Check Flex combines laser and inkjet printing technologies on up to three printing sides. With line speeds up to 300 cartons per minute, the Print & Check Flex is designed to handle cartons spanning from “match box” to “shoe box” with weights up to 500 grams.
Anti-counterfeiting, gentle handling offer protection
Schreiner MediPharm (schreiner-medipharm.com) introduces the next generation of its popular Flexi-Cap, an innovative security solution that irreversibly indicates the first opening of primary containers to prevent their illegal reuse with counterfeit substances. The new Flexi-Cap Plus features a tear strip running through the label as an integrated element. The label is destroyed upon initial opening and cannot be reused as an alleged original, offering more protection against tampering.
With additional security technologies – like holograms, color-shifting inks, void effects or security pigments – further hurdles to tampering can be raised. Flexi-Cap Plus can be customized to accommodate various glass container types, shapes and sizes, and can integrate into a pharmaceutical manufacturer’s existing brand design. The lid of the film cap also offers additional space that can be used for imprinting barcodes or integrating NFC chips for interactive applications.
The Model SP-29 case packer by A-B-C (abcpackaging.com) responds to the need for gentle handling of shrink-wrapped pharmaceutical and other hard-to-pack medical products. A-B-C developed the packer to automatically accumulate the entire case load in precise position before sliding it neatly into the case. This unit packs cases more carefully than hand labor and is built for efficient, delicate movement throughout the packing process. And with manual packing eliminated, only one operator is needed to open and position the case.
A-B-C’s stainless steel accumulation table and packing station assure smooth transfer of many package surfaces and shapes. A pump-up system with an innovative collating mechanism accumulates each load, elevates and orients it accurately into the predetermined pack pattern, and then automatically slips the load into the case and lowers it to be conveyed to sealing.
Blister packaging enhances shelf life
Oral solid doses can face a packaging headspace challenge that impact shelf life; besides moisture and oxygen ingress, exposure to certain conditions can lead to the formation of volatile organic compounds (VOCs) and other gases that, in turn, may interact with drug products and adversely impact their stability.
CSP Technologies’ (csptechnologies.com) patented engineered active polymers, utilized within pouch and blister packaging, may be an effective and efficient headspace management solution, addressing a wide range of desiccant and scavenging needs without the use of adhesives or purging. CSP’s Activ-Blister™ solutions help control the internal atmosphere of existing individual blister cavities, allowing for improved product performance and enhanced shelf life. Offering moisture, oxygen and combination absorption, the technology can be applied without the use of adhesives and without changes to the existing footprint of a packaging line. Activ-Blister can be incorporated into a wide range of blister packaging formats.
New and innovative pharmaceutical packaging solutions will prove to be a driving force for the market. The key to the success of any company is to be aware of the trends and new technology at all times in order to keep ahead of the competition. Being the first – and being the best – is crucial.
Mayo Medical Laboratories implements automated specimen system
The automated laboratory, in Rochester, MN, handles tens of thousands of patient vials every day from more than 3,000 tests across the full spectrum of clinical laboratory subspecialties. Laboratory leadership wanted to continue to improve turn-around time from when specimens are received by Mayo Medical Laboratories internal operations to when the specimens arrive at the one of 60 clinical laboratories for testing. The operations team also wanted perfect accuracy when directing specimens through pre-analytic workflow.
As the Mayo Medical Laboratories test volumes continue to increase, the staff needed robust automation to meet aggressive productivity goals. The previous automation line was limited to handling from 2,500 to 3,000 vials per hour and required a certain level of manual pre-sorting. While one-in-one-million quality defects is acceptable in some types of manufacturing, this level of inaccuracy is unacceptable for a patient care provider and commercial reference laboratory handling one million patient specimens every week.

A laboratory assistant places ambient specimens onto the Mayo Medical Laboratories automated sortation system. (Photo courtesy of Mayo Medical Laboratories)
Engineers from Yaskawa Motoman (motoman.com) and a team from Mayo Labs evaluated potential solutions for addressing the laboratory’s goals and then settled on a new installation in August 2014. The solution included a large track-based system with six configurable Yaskawa AutoSorter 1200T output sorters. The new line also included six container scanning system (CSS) stations, which read specimen labels and link them to radio-frequency identification (RFID) tags on conveyor pucks. Once the CSS establishes the bond between specimen and tag, the station sends the specimen to the main conveyer and directs it to the proper destination.
“Laboratory personnel can place specimens right on the new line, and we can route 80% of all specimens through automation,” says Aaron Maixner, operations manager for Mayo Medical Laboratories. “Prior to August 2014, only 60% of the daily volume was processed via automation.”
Higher throughput is from the system’s ability to sort 6,000 specimens per hour, a 100% increase over the prior line’s speed. The line can also sort vials into 540 targets versus the previous system, which topped out at approximately 320 targets.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!




