Automation
7 Tips for Choosing Cleanable Packaging Equipment
Machines incorporating sanitary design can improve the bottom line by lowering the risk of product recall and improving productivity.




Sanitary design and cleanability offer opportunities for improving the bottom line. Complying with Current Good Manufacturing Practices (cGMPs) for equipment and utensils lowers the risk of expensive product recalls and the consequent damage to brand reputation. Designing equipment to be cleanable ensures that the equipment and utensils can be cleaned faster. Less time cleaning means more time producing, which improves overall equipment effectiveness (OEE) and ensures that more product ships. Replacing older equipment with new, clean design automation provides a competitive edge in terms of higher quality, less downtime and greater flexibility.
The Food Safety Modernization Act (FSMA) defines requirements for food equipment under CFR Title 21 117.40. Paragraph A states that “All plant equipment and utensils used in manufacturing, processing, packing or holding food must be so designed and of such material and workmanship as to be adequately cleanable and must be adequately maintained to protect against allergen cross-contact and contamination.” How does cleanability influence sanitary design and what cGMPs must OEMs follow to achieve the standards listed in CFR Title 21 117.40?
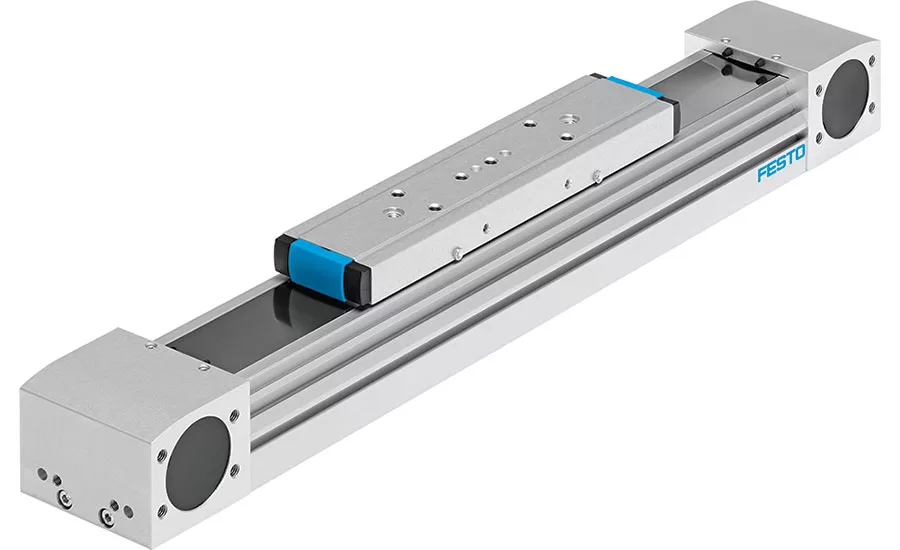
The electric tooth-belt actuator offers an example of how 117.40 impacts design for components applied outside of food and splash zones. This actuator has an easy wipe-down exterior. Its profile prevents build-up of dirt or debris. The unit’s body can be pressurized to help prevent unwanted ingress of particles.
Here are seven tips on what to look for and why in cleanable equipment and utensils. These suggestions are based on section 117.40.
Tip 1: Easy to Clean
Small radii and corners pose a hygiene risk because flow velocities of the cleaning agents and disinfectants are substantially reduced in tight spaces and the required cleaning effect isn’t achieved. OEMs should have a minimum radius of 3 mm on parts.
A high-gloss surface finish is essential on components that come into contact with the food product in order to reduce microbial contamination. This can be achieved by using a mean peak-to-valley height of 0.4 to 0.8 µm within the food zone. High-quality surface finishes and large radii, such as those found on the pneumatic actuator, make cleaning the actuator quick and easy. FDA-approved seals and NSF-H1 grease protect against unintentional adulteration of the food product.
Tip 2: Designed for the Cleaning Process
All machines and system components must be designed to eliminate dead spaces. Product remnants in dead spaces are difficult, if not impossible, to remove. Therefore, machinery and components must be designed to be either completely open or completely sealed.
Cleaning processes often employ the use of high-pressure hoses. Equipment and components must be designed to withstand these processes. Resistant surfaces and a high IP protection class, such as the IP69K-rated valve terminal, are designed to operate effectively in harsh environments.
Tip 3: Corrosion-Resistant Materials
Many potential sources of contamination in food production, such as bacteria, chemical influences or corrosion particles, can be eliminated by utilizing a few basic design considerations. To ensure that cleaning is safe, the materials used must not react with the cleaning agents or the disinfectants. Machine parts must be resistant to corrosion and be mechanically and chemically stable.
Its design rigorously conforms to cGMP criteria. For example, there are no threads on the bearing cap and, thus, a reduced possibility of trapping contaminants. Its self-adjusting end position cushioning system is designed without contaminant-susceptible adjusting screws.
Tip 4: Proper Component Selection to Withstand Cleaning Processes
Plastics and elastomers permitted to come into direct contact with food must comply with the directives of the FDA. In addition to resistance to stress, ease of cleaning is also an important factor in the selection of suitable plastic materials. The material must not give off or absorb any hazardous substances.
Tubing is at risk from various environmental influences as shown by the microscopic crack in the micrograph below.
Proper components selection to withstand cleaning processes will resist cracking.
Approximately 90% of the defects on pneumatic tubing are traced to chemical, microbiological or physical influences. Proper tubing selection can minimize or eliminate failures due to these influences. PUN-H and PTFEN tubing are ideally suited for use in the food industry. Both are resistant to cleaning agents, are FDA compliant, and are resistant to microbes and hydrolysis. PUN-H is more flexible and economical, while PTFEN is ideal for the harshest environments.
Tip 5: Proper Lubricants
The U.S. has the strictest regulations on the use of lubricants and additives used in the food industry. Lubricating greases and oils must comply with CFR 21 178.3570. For equipment and components that will unavoidably come into sporadic contact with foods and primary packaging, approved lubricants such as NSF-H1 must be used.
Intensive cleaning of machine parts can also wash out the lubricating grease and impair the operation of components. Using dry-running seals ensures that the washed-out machine components still function reliably. Eliminating the use of components that fail because lubrication can be washed away is an example of how cleanability and cGMPs can improve uptime.
Tip 6: Eliminating Exposed Threads
Threads provide the perfect breeding ground for contaminants as the small spaces between a metal-to-metal contact cannot be cleaned. Any threads that cannot be avoided should therefore be closed off with suitable blanking caps and then sealed. Clean design means eliminating exposed threads.
The R and NPT seal by wedging threads together and through the use of sealing tape. These tapered threads risk contamination, not only through exposed threads, but also through metal fragments or flaking tape. Parallel G-thread seals flush to a gasket and eliminates exposed threads. Utilizing G-threads wherever possible is an easy choice for improved FSMA compliance.
Tip 7: Compressed air quality
CGMPs under 117.40 do not define a minimum pneumatic air quality for food and primary package contact, which means the required filtration level must be determined by the end user. While many end users have determined their own air quality requirements, others may be unsure of recommended purity. Organizations such as BCAS, 3A, VDMA and SQF have published recommendations for specific air quality requirements. SQF guidance documents, for example, recommend a final filtration stage in food contact areas of 0.01 micron with an efficiency of 99.999%.
The compressed air preparation unit is available for various air quality requirements and flow ranges. The pictured example has a filtration cascade ending in a 0.01 µm filter with 99.9999% efficiency. Assuming 7:4:4 air quality from the compressor, the resulting air quality of 1:4:2 meets the ISO 8573-1:2010 standard for air purity.
It is important that OEMs have robust clean design component specifications and that OEM purchasing personnel understand why these specifications are in place and must be adhered to. It is incumbent on OEM sales personnel to educate their customers on cGMPs, why these practices are in place and their associated benefits.
Festo has downloadable educational materials on sanitary design that both OEMs and end users can benefit from studying. Links to these materials can be found at festo.com/foodsafety.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!



