Home » Keywords: » serialization
Items Tagged with 'serialization'
ARTICLES
Cover Story
A Prescription for Pharmaceutical Packaging
Serialization, safety and security head a long list of challenges for the pharmaceutical packaging industry in 2018
April 1, 2018
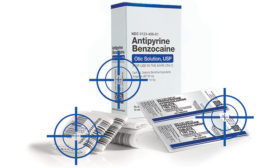
Machinery Technology: Printing & Coding
The challenge of barcode grading in a serialized world
Barcode grading assists in maintaining a standardized level of quality and operability.
October 6, 2017
Cover Story: Healthcare Packaging
Healthcare packaging solutions
Serialization and patient adherence breakthroughs.
June 13, 2016