Home » Keywords: » compliance packaging
Items Tagged with 'compliance packaging'
ARTICLES
Materials Technology: Blisters & Cards
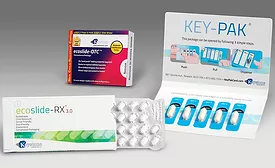
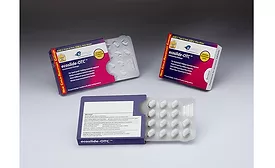
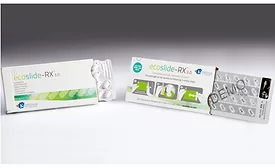
Blister Packaging vs. Bottles for Pharmaceutical Products
Factors to consider for packaging over-the-counter and prescription medications include safety, adherence and product quality.
May 15, 2018
Cover Story
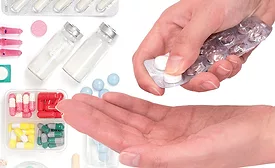
Pharmaceutical packaging must perform to work
How performance and functionality impact the pharmaceutical packaging segment.
April 12, 2017
Healthcare Compliance Packaging Council Column
Let’s make a New Year’s resolution to improve pharmaceutical safety
February 13, 2017