Cover Story
Pharmaceutical packaging must perform to work
How performance and functionality impact the pharmaceutical packaging segment.





The medical and pharmaceutical industry is growing. It’s one of those segments that will always be here and most likely continue to grow due to more disease, better prevention medications and an aging population. According to PMMI, The Association for Packaging and Processing Technologies, the market for pharmaceuticals and medical devices does, in fact, continue to grow. In total, the 2015 pharmaceutical industry produced $1.1 trillion in global sales with medical devices reaching $350 billion. An aging population, longer life expectancy and a general increase in rare or chronic diseases is impacting healthcare worldwide. Global medicine use is expected to reach 4.5 trillion doses by 2020, up 24% from 2015, emphasizing the opportunity in this market segment.
That’s all fine and dandy, but the key to a product’s success lies in the packaging. The medicine or device may be a home-run product, but if consumers aren’t using the product correctly, it has little chance of success. And if the consumers cannot even open the package to get to the product inside, it has zero chance.
Senior-friendly isn’t just nice to have; it is a must for pharmaceutical companies to create packaging to cater to this growing population who struggle with opening their medications. Senior-friendly packaging refers to packaging that has been specifically created for the convenience of senior-aged people. These packages include easy to open and seal, visually detectable, visible dosing, and labels that clearly communicate the benefits of taking the product and the repercussions of skipping the prescribed dosages. According to Future Market Insights (futuremarketinsights.com), between 2010 and 2050, the population of seniors in America is expected to increase significantly.
Scott Lucas, managing director, Brandimage North America (brand-image.com) says that compliance packaging is the key to satisfying this demographic.
“Pharmaceutical companies providing medication for the elderly find that compliance, that is, the extent to which patients follow provider recommendations for day-to-day treatment with respect to timing, dosage and frequency, is the number one issue impacting packaging design. Studies have found that packaging that reminds people whether they have taken their medication, e.g. certain antibiotics, steroids and hormone replacement, increases compliance significantly among the elderly. The estimated annual cost the U.S. incurs as a result of poor medication compliance approaches $300 billion as recently noted in the New England Healthcare Institute paper “Thinking Outside the Pillbox,” 2010. Poor compliance is the primary cause for 125,000 deaths annually (342 people every day) and an estimated 10%-25% of hospital and nursing home admissions. Beyond the financial impact on the healthcare system there is a risk to the brand value, perception and performance. When a treatment fails or underperforms due to a lack of compliance the patients never blame themselves, they place the blame on the medication, hurting the perception of the brand. When that patient discusses the lack of performance with their medical provider, again they often aren’t aware of their lack of compliance or won’t admit to it – thus the medical provider also can assign blame on the medication and brand.”
Compliance packaging solves this problem and then some.
“It’s clearly in the best interest of the drug manufacturers and their brands to create packaging that facilitates compliance to ensure a positive brand experience. Some of the added features on packaging that can help are dosing indications and reminders, patient specific information, easy to open, close and manage form factors, and clear and plain typography and messaging,” continues Lucas.
If it’s important for packaging to be geared toward seniors, and really any generation that experiences packaging frustrations, it is perhaps even more important to keep that packaging difficult to open for children. But this can be a difficult balance to achieve.
According to Future Market Insights, in 2014 as per U.S. medical data, three out of four cases of child drug poisonings, the medication belonged to a parent (39% of cases) or grandparent (38%). Because of this danger, there is increasing demand for packaging to be equally senior-friendly as it is children-resistant.
“The advantage of the childproof cap on bottled medications is that children find it very difficult to open; conversely, these caps are also difficult for many adults to open, especially among the elderly. Creating patient/consumer needs that are at odds with each other forces a brand to choose between ease of use for the elderly or safety for children. That’s why the pharmaceutical industry is packaging more and more oral medications in blister packs, with quantities varying by prescription. Also, new childproof caps are available that provide safety and ease of access when clear “opening” directions are provided. Medications can provide the same level of protection through complex closures if they can provide both clear instructions for adults to manage them and account for larger hands that need to gain access and smaller hands that are being denied access,” continues Lucas.
What’s senior-friendly, child-proof and keeps pharmaceutical users on track with their mediations? The MedLock EZ family of patient compliance packages from Colbert Packaging Corporation (colbertpkg.com).
MedLock EZ consists of a fully brandable outer paperboard shell housing a pill blister card that is locked into place with an integrated locking mechanism. The lock disengages by squeezing and holding indicated touch-points on one end of the shell and sliding out the blister pack through the other end. The medication can then be removed from the blister pack and slid back into the carton, which then locks it into place.
MedLock EZ is a secure and sustainable unit dose alternative to amber pharmacy vials. Subjected to Consumer Product Safety Commission (CPSC) testing protocols, MedLock EZ has tested to F1, which is a testament to the effectiveness of its child-resistant, senior-friendly design attributes. MedLock EZ is available in multiple footprints to accommodate a wide range of medications.
“When subjected to CPSC testing protocols, all of the seniors were able to open the MedLock EZ package, but none of the kids were able to scratch, tear or bite their way in,” says Glenn Grosskopf, vice president of product development for Colbert Packaging. “There really is no other acceptable outcome to the CPSC tests. MedLock EZ is a child-resistant, senior-friendly and sustainable unit dose alternative to amber prescription vials, and now it’s available in two varieties.”
MedLock EZ’s large printable area offers the opportunity for pharmaceutical companies to engage their customers and promote compliance through design features like clear and consistent dosing instructions and by making dosages easy to dispense and track.
A similar paperboard/ blister combination pack from Multi Packaging Solutions (MPS, multipkg.com) offers the flexibility and customization to suit product type and machine filling requirements. The company has expanded its range of child-resistant packs with a wide range of design options and configurations available up to the highest F1 child resistant rating.
The new range of packs visually resembles standard folding cartons but is made from tear-resistant paperboard. They incorporate unique locking features that help to prevent children from accessing the pack’s contents while still allowing senior consumers easy access. For convenience and ease of use, the pack remains together as one-piece when opened and will automatically re-lock when closed.
“It’s important for brands to help safeguard consumers against unintentional accidents by dispensing powerful medicines in packs that restrict access to children whilst offering clear communication for patient compliance. These packs can incorporate compliance features within the artwork and are compatible with many different products including blisters, tubes and vials,” says Bobby O’Connor, senior vice president sales – Global Healthcare, MPS.
One company is getting ready to shake up the industry with its one-piece child-resistant cap which only requires a quarter turn to apply or remove. Comar (comar.com) has developed its new SecureCap® QuarterLoc one-piece push and turn bottle closure for application in the nutraceutical, over-the-counter (OTC), pharmaceutical and other CPG markets. The cap is easier to use and more intuitive than the traditional two-piece closures consumers are used to. Designed to be compatible with multiple products, the closure has passed CPSC testing protocols for both child-resistance and senior-friendly performance. Stock sizes will be built based on demand and commercial production is expected to commence later this year.
By providing a secure closure with just a quarter turn, QuarterLoc is easier for patients to use and keeps the product more secure, as it eliminates the potential of thread misalignment and cocked caps.
“The primary package (bottle and closure), including the associated dosing/dispensing device, needs to be convenient and easy-to-use but also safe and reliable. The challenge for packaging designers and manufacturers like Comar is to provide the most aesthetically pleasing, convenient, affordable package that also ensures product security and freshness and complies with the CPSC Protocol for child resistant, senior friendly packaging. In our view, the packaging is an extension of our customers’ brands so we’re constantly trying to be innovative with regard to form, fit and function so that the ultimate end user gets the most value out of each and every purchase,” says John Daly, director of technical operations at Comar.
For nutraceutical, OTC and prescription medicine manufacturers, there have been few innovative design changes that adhere to compliance with the CPSC requirement for child-resistant packaging based on anticipated qualification delays and costly retooling of filling processes. However, the design of the SecureCap® QuarterLoc CR closure offers a cost effective solution since it is easily integrated into existing packaging lines. In addition, the QuarterLoc offers a reduction in part weight, assembly steps and production costs.
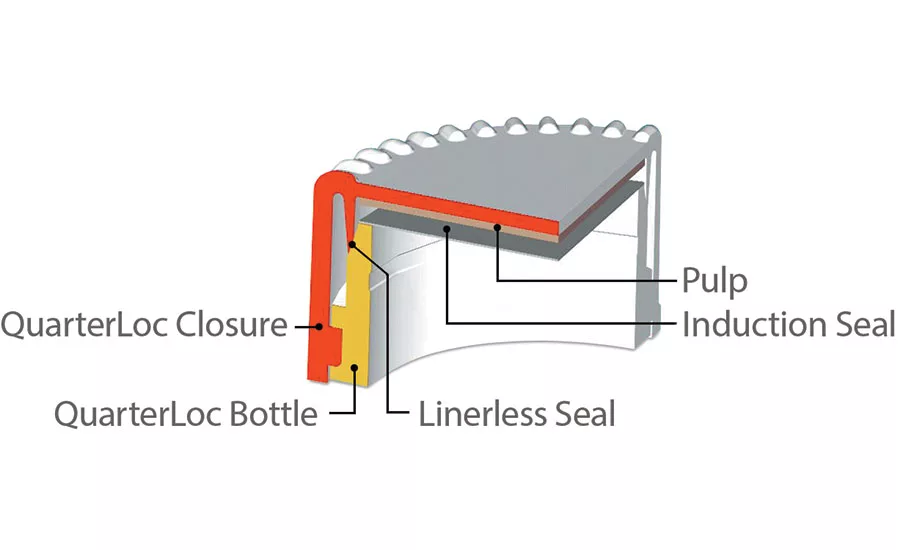
Comar has also developed bottles with complementary neck finishes for use with QuarterLoc closures. SecureCap® QuarterLoc is manufactured from polypropylene and works equally well with HDPE or PET bottles. The one-piece design reduces manufacturing complexity and is more environmentally responsible. The closure does not require a liner but can accommodate a range of foil induction liners or in line conduction liners.
Pharmaceutical packages must be functional to perform correctly. Compliance and senior-friendly packaging helps keep consumers accountable, while child-resistant helps keep non-consumers, i.e children safe. When working in tandem, these packages deliver multiple benefits for many groups and help ultimately in keeping patients safe.
|
Induction cap sealing addresses tamper-evident concerns in healthcare packaging
|
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!

 by John Brown, vice president, marketing of Selig Group
by John Brown, vice president, marketing of Selig Group Selig (
Selig (

